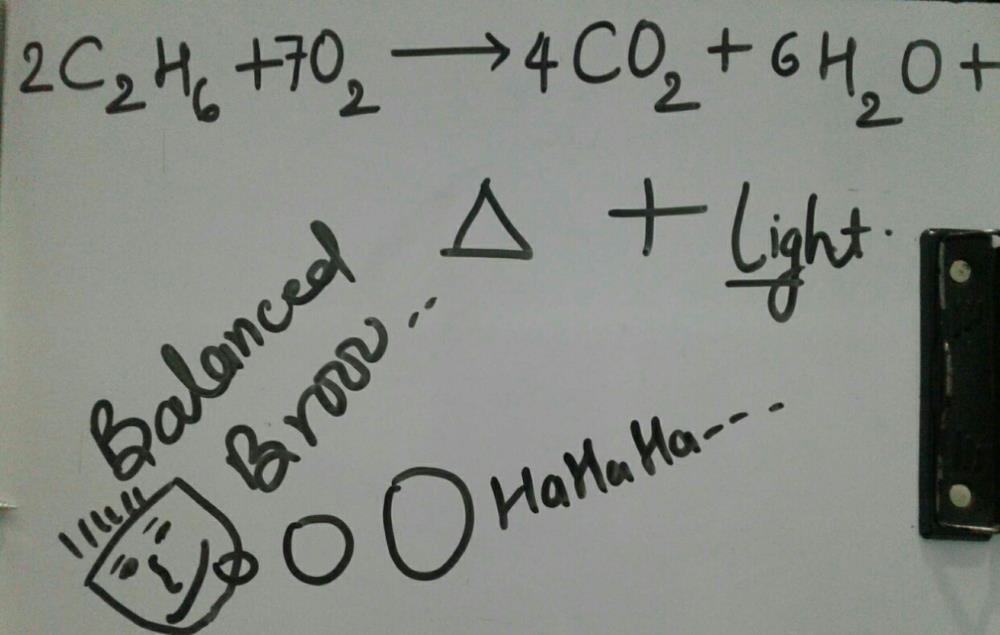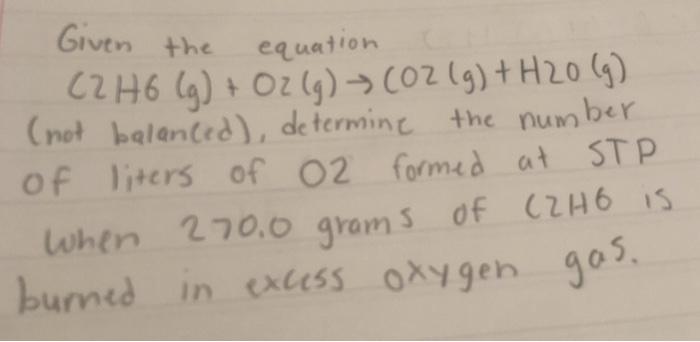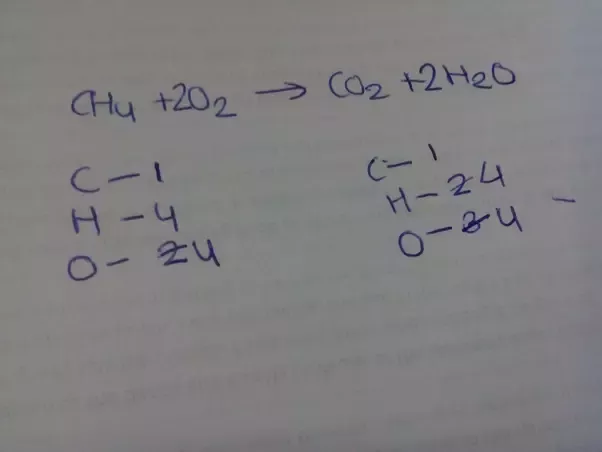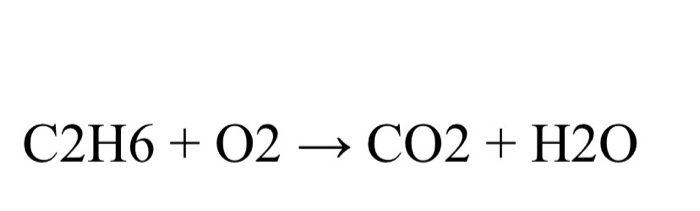Balance the following equation by oxidation number method. C6H6 + O2 → CO2 + H2 O - Sarthaks eConnect | Largest Online Education Community
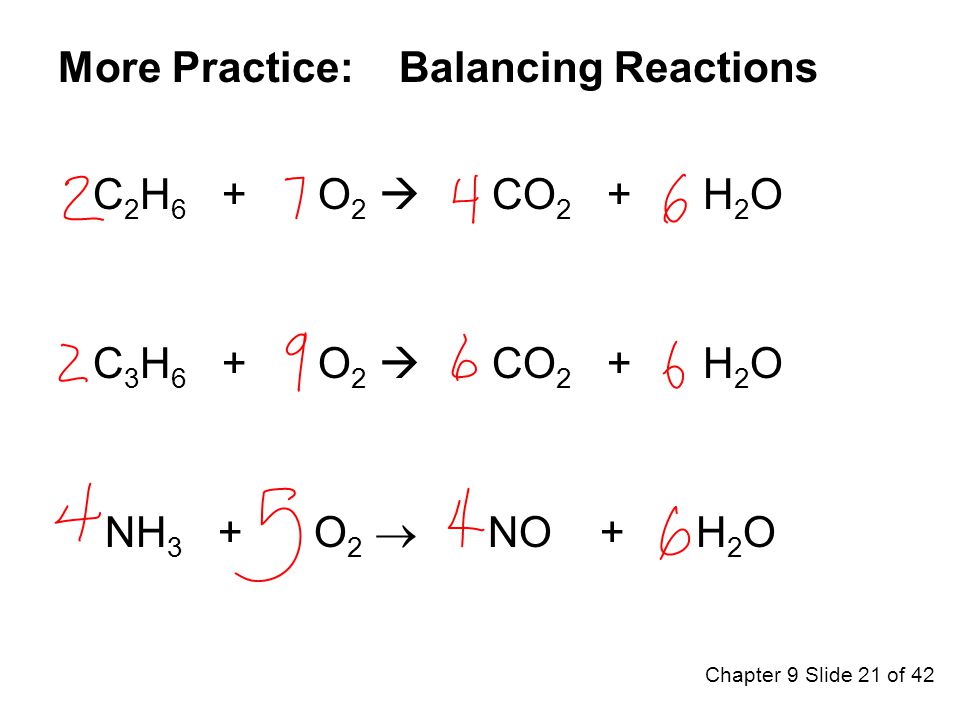
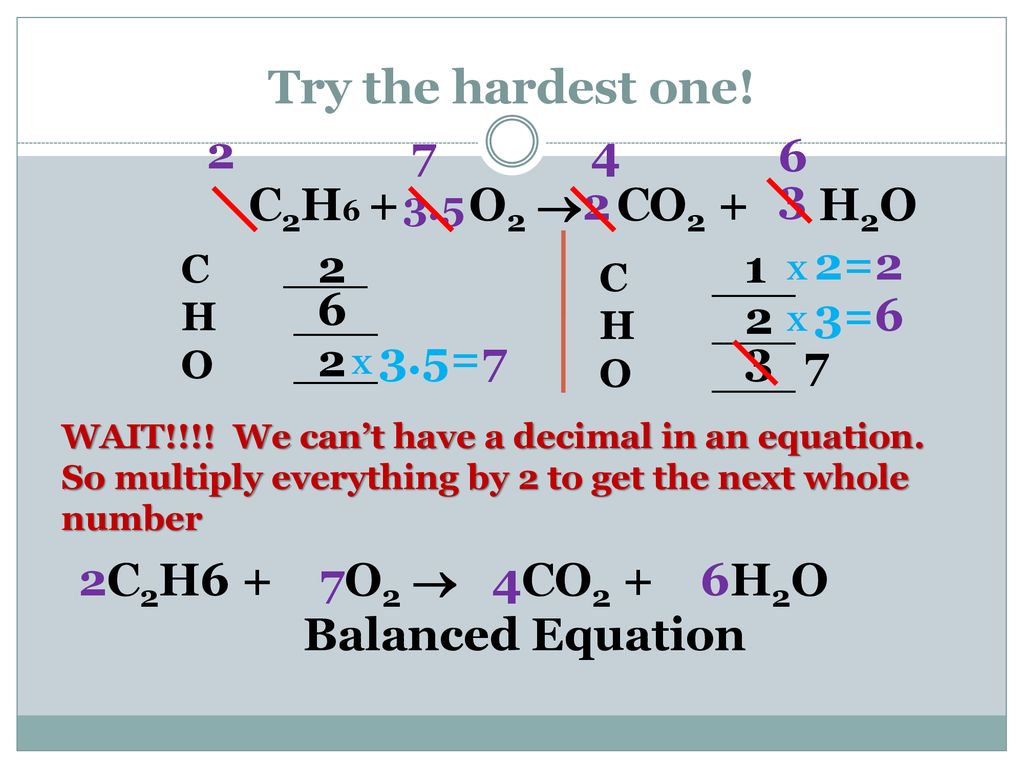
By using the Gaussian elimination method, balance the chemical reaction equation: C2H6 + O2 → H2O + CO2 - Sarthaks eConnect | Largest Online Education Community

C6H6 + O2(g)→ CO2(g) + H2O(g) When the equation for the reaction represented above is balanced and all coefficients are reduced to lowest whole number terms, the coefficient for H2O(g) is :

Chapter 6: Writing and Balancing Chemical Equations. AB A + B. CaCO3 CaO + CO2 A + B C. AB + C AC + B (or AB + C CB + A) - PDF Free Download

C2H6 +O2 =CO2 + H2O Balanced Equation C2H6+O2 combustion(Ethane) Balanced reaction and Equation - YouTube